
The National Agency for Food and Drug Administration and Control has been informed by Glaxosmithkline Pharmaceutical Nigeria Limited on the circulation of falsified Zinnat Suspension a brand of Cefuroxime Axetil Lot Number 349R in retail outlets in Nigeria. This lot number (349R) has been used on the genuinely manufactured Zinnat Suspension 125mg/5ml which expired in MAY 2020.
The details of the falsified Zinnat Suspension 125mg/5ml brand of Cefuroxime Axetil is as shown below;
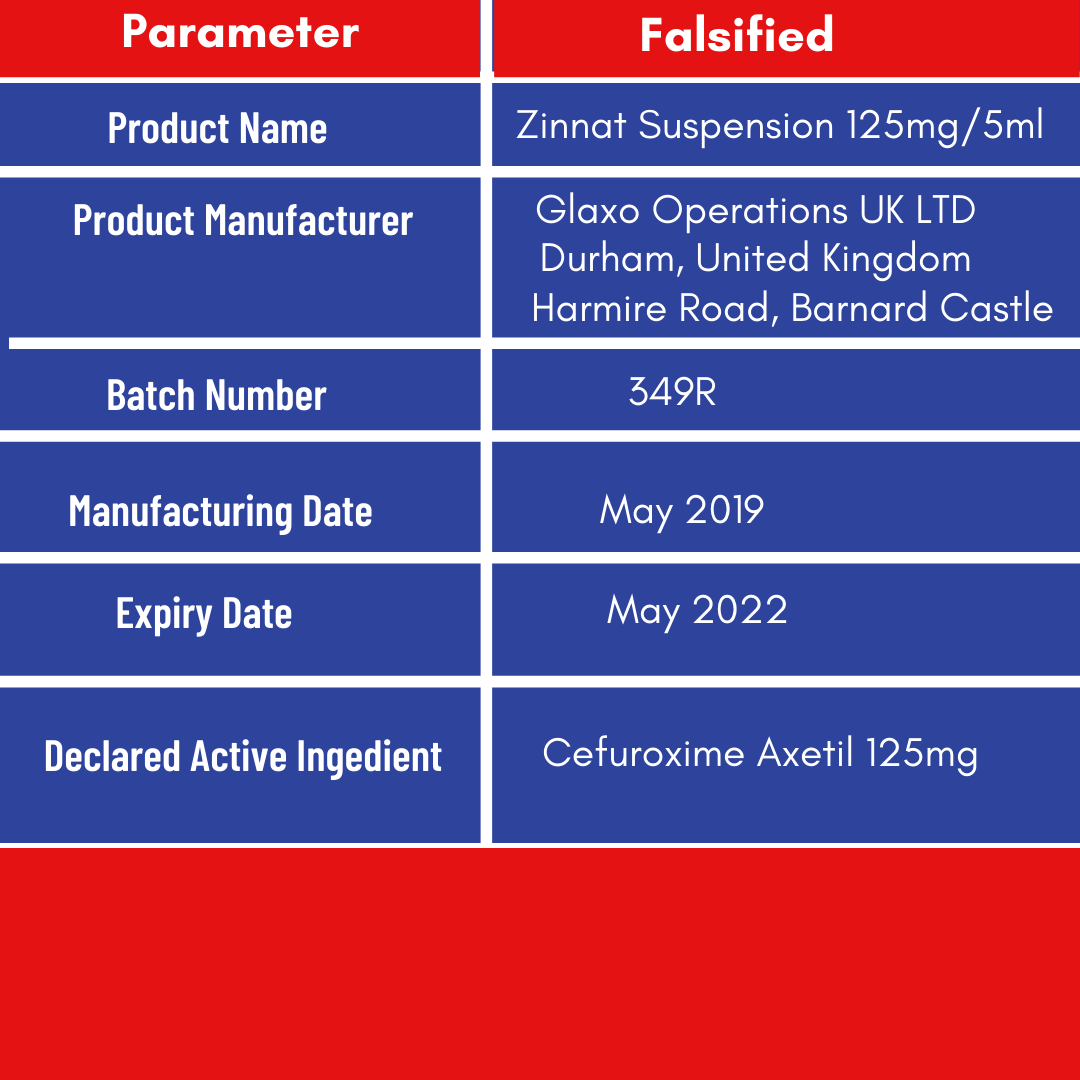
The genuine Zinnat Suspension 125mg/5ml (Cefuroxime Axetil 125mg/5ml) Batch Number 349R manufactured by Glaxo Operations UK Ltd has Manufacturing date MAY 2018 and Expiry date MAY 2020.
The genuine Zinnat Suspension 125mg/5ml manufactured by Glaxo Operations UK Ltd is registered by NAFDAC with NAFDAC Reg. No. 04-0463.
Glaxo Operations UK Ltd has confirmed that they did not manufacture the affected falsified Zinnat suspension 125mg/5ml.
Please Stay Alert.
Part of our mission at RxAll is to fight counterfeit drugs and create awareness to the general public on ways to stay off these fake drugs. We would continue to fight and provide access to safe and Authenticated drugs through our online drug delivery platform.